It is controversial, diverse and potentially revolutionary. But what do we know about it?
Though much has been made of treatment involving platelet-rich plasma – from efficacy to formulations, hype, uses and potential – relatively little data exists to firmly support or refute its capabilities.
The very nature of platelet-rich plasma (PRP) is one problem: As the autologous PRP for every patient is different, it is difficult to create any one particular formulation for study that would properly apply to all patients. These differences pose a large problem for physicians and scientists, who have found that the work is not necessarily in the study of PRP but rather the development of the studies.
Furthermore, said Steven P. Arnoczky, DVM, since PRP is being used in so many ways on such a wide variety of pathologies, consistent results have been difficult to acquire.
These are only some of the problems surrounding the further development and use of PRP, and while answers may be on the way, many questions are lingering.
How can PRP be defined?
Though the efficacy of PRP is possibly the largest point of contention among physicians, some suggest the larger question is that of what is PRP, actually.
“The lay press does not understand the complexity of PRP,” Allan Mishra, MD, said. “As more data emerge, it will become clear to both physicians and surgeons that the specific formulation of PRP matters significantly in terms of bioactivity.”
“The thing I would emphasize is that we have got to get a better understanding of the formulations in order to figure out whether PRP is valuable or not,” he added. “What is missing in the lay press – but also in the medical literature – is the discussion of what PRP is.”
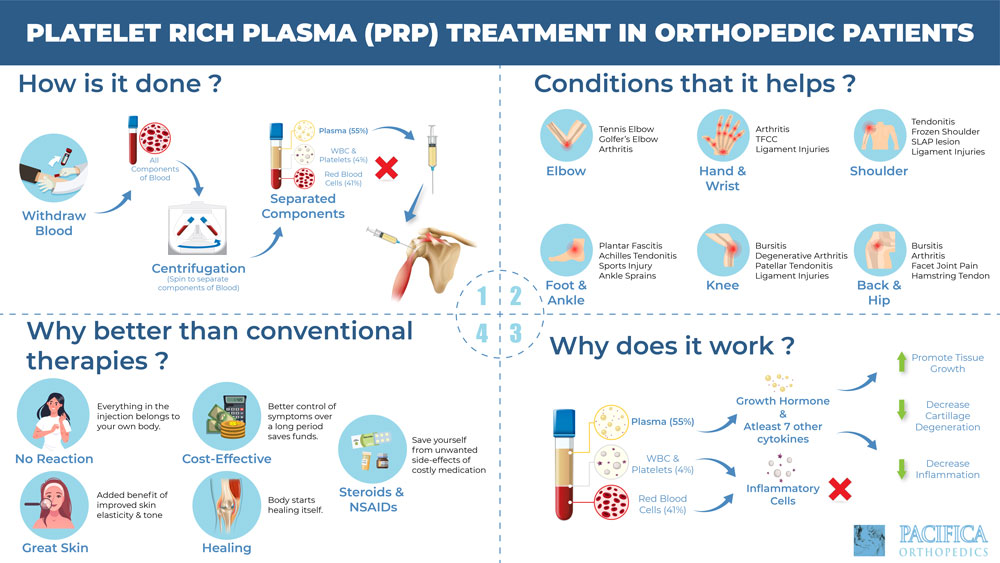
Mishra pointed out that a formulation of PRP containing only concentrated platelets differs greatly from a formulation composed of concentrated platelets and white blood cells.
Arnoczky said that indications, benefits and limitations of PRP would be best determined through extensive trial, but stressed that such studies are “an onerous task” due to PRP’s lack of definition. Among the issues, he said, are:
- variations in preparation techniques: platelet harvest efficiency, the presence or absence of white and/or red blood cells, the use of thrombin to activate platelets;
- the biologic variability among donors, such as individual variation in platelet count; and
- absence of a standard dosing protocol: Is it 1cc or 2cc of a given PRP preparation administered? What level of cytokine concentration does that present?
Nicholas A. Sgaglione, MD, said that while the definition has been a hurdle in the creation of efficacy studies, it is a problem that could soon be solved.
“It is very variable, so investigators are not always clear about what to study and how to study it,” he said. “Dosing, application, schedule, two injections vs. one – all of that produces a problem. I think we are already in the phase where that is changing and the data will start to accrue.”
What is the best use for PRP?
Thus far, physicians are finding that the best indication for the use of PRP is tendinopathy. According to Mishra – who published a study in 2006 which found a 93% reduction in pain after 1-year follow-up for patients of chronic lateral epicondylar tendinopathy treated by PRP – forms of chronic severe tendinopathy in the Achilles, patellar and quadriceps could be sufficient indications.
“A study of partial rotator cuff tears would also be interesting,” he added.
Sgaglione said that he is “very limited and narrow” in terms of how he uses PRP.
“I have selected and chosen to treat patients with isolated meniscal tears who are biologically at risk for failure of meniscus repair,” he said, adding that he places the PRP arthroscopically using a fibrin-rich matrix to add to the meniscus repair construct and augment or increase the healing rates of those tears.
Though PRP has shown promise in the area of tendinopathy, Mishra noted that it should not be seen as the go-to treatment without more data to back up its use.
“Patients with mild or moderate tendinopathy that has not been treated with other means such as rest, physical therapy and activity modification should not be given PRP unless they fail these modalities,” he said.
What defines a good result?
Arnoczky, an Orthopedics Today Editorial Board member said one major issue with determining the efficacy of PRP is that of physicians expecting “the home run” – clear, large-scale improvements in functional outcome.
“I think in contrast to other orthopedic therapies like [total knee arthroplasty] or [total hip arthroplasty] where functional outcomes are more ‘clear cut,’ clinical studies looking at the efficacy of biologics like PRP in heterogeneous populations such as tendinopathy and rotator cuff repair will require a more nuanced study design,” he said.
Arnoczky added that biologic agents could bring forth an early response that becomes less pronounced over the long-term, or vice-versa.
Freddie H. Fu, MD, MD, DSc, DPs. (Hon), cited mixed results from several studies that questioned the value of PRP treatment as compared to alternatives – or, in some cases, placebos.
“[In the de Vos study], the use of PRP in Achilles tendon injury was compared with regular saline injection and no difference could be found,” he said. “Another study by Peerbooms et al. compared platelet injections with injections of a steroid in patients with tennis elbow and 73% of the platelet-treated group saw improvement at 1 year, compared with 51% with the steroid injections. But can we consider 73% a real ‘success’?”
According to Sgaglione, improvement may not be a question of huge gain so much as incremental success.
“Augmentation of healing is the key phrase,” he said. “If you have certain tissues that heal 60% of the time, and you are able to increase it by 10% to15%? That is significant.”
Evaluating the improvement offered by this therapy is also tricky. “We need to go beyond ‘do you feel better’ to ‘are you really better,’” Mishra said. He added, ultrasound evaluation of tendon or ligament thickness can be used as an objective measure of improvement, as well as the monthly evaluation of hypoechoic signal as compared to the patient’s pain scores and functional outcome.



0 Comments